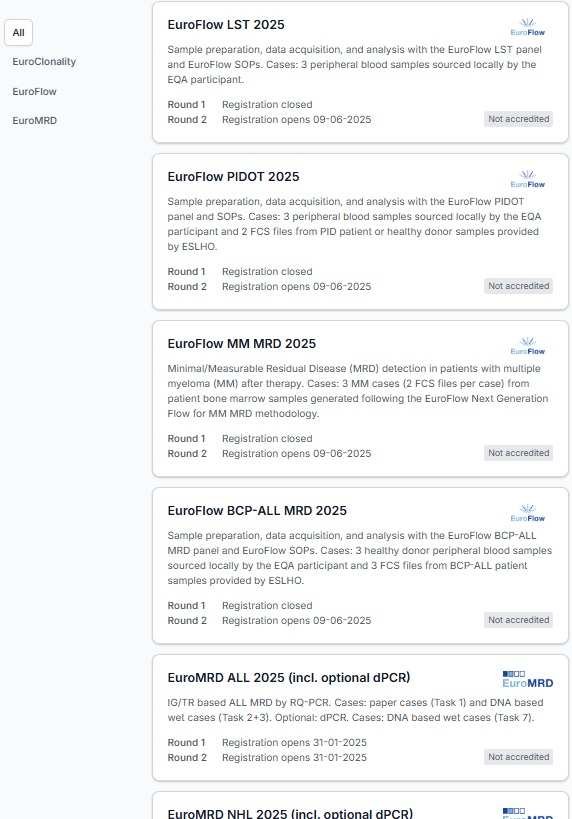
EuroFlow BCP-ALL MRD 2025
B-cell Precursor Acute Lymphoblastic Leukemia Minimal Residual Disease (BCP-ALL MRD) EQA scheme
Aim of the scheme
The BCP-ALL MRD scheme is intended for laboratories that want to assess their performance in utilizing EuroFlow’s BCP-ALL MRD antibody panel (van Dongen et al. 2012, Theunissen et al. 2017) and relevant EuroFlow standard operating procedures (SOPs) in their routine diagnostics as well as their ability to analyze and interpret flow cytometry standard (FCS) files obtained through this methodology using BCP-ALL patient samples.
The scheme is divided into two parts. In the wet lab part, you will stain 3 locally sourced healthy donor peripheral blood samples with the BCP-ALL MRD Tube 1 antibody panel and analyze the data. In the dry part you will merge the files you obtained in the wet part with 3 BCP-ALL patient FCS files provided by ESLHO and analyze them together for MRD. The provided FCS files are generated by a EuroFlow reference laboratory using the BCP-ALL MRD Tube 1 antibody panel and following the EuroFlow methodology. For both parts, you will report your results and conclusions via the ESLHO EQA Portal.
Your results will be compared to the consensus reference interpretation, which is defined by three experts. Additionally, your results will be compared to the other participants in the round.
Two unique rounds of the BCP-ALL MRD scheme are offered in 2025: one in spring and one in autumn.
Performance and report
After each round, participating laboratories will be provided with an EQA certificate showing their performance as compared to the reference values and to other laboratories. In addition to the performance report, specific comments or recommendations on how routine practice could be improved may be provided.
Further details on data analysis, how participants’ performance is evaluated, and on the contents of the report can be found in the document “Scheme information” available on the Resources page.
Educational meeting
All EuroFlow schemes’ EQA rounds offered in 2025 will be concluded with an educational meeting, which will include all rounds performed throughout the year. Participation is free of charge for two representatives per participating laboratory. During the meeting, the rounds’ results will be shown (anonymized), possible problems and pitfalls will be discussed, and there will be the opportunity to receive direct feedback. More information regarding the educational meeting will be announced on the ESLHO EQA Portal.
Organization
The BCP-ALL MRD scheme is organized by ESLHO in collaboration with the EuroFlow EQA Committee. The EuroFlow EQA Committee is composed of members of the EuroFlow Consortium (www.euroflow.org). The laboratory at the Charles University, Prague, Czech Republic, operates as the lead expert laboratory, with Michaela Reiterová, MD, PhD in the role of lead subject-matter expert.
Further details on the task division of subject-matter experts can be found in the document “Scheme information” available on the Resources page.
Participation
ESLHO offers the BCP-ALL MRD 2025 rounds free of charge to participants of the EuroFlow Consortium.